 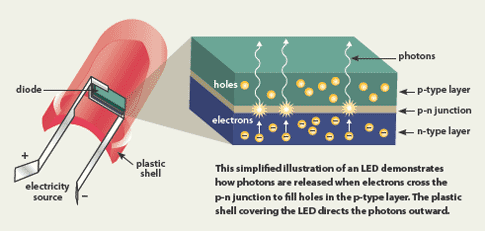
LEDs offer a variety of advantages to lighting professionals and ultimate beneficiaries of LED lighting systems - from creative individuals to innovative businesses to visionary cities and countries: Different materials produce photons at different wavelengths, which appear as light of different colors. The material in an LED is selected so that the wavelength of the released photons falls within the visible portion of the light spectrum. When extra electrons in the n-type material fall into the holes in the p-type material, they release energy in the form of photons - the basic units of electromagnetic radiation.Īll diodes release photons, but not all diodes emit light - just light-emitting diodes. 
When they get close to each other, the n-type atoms donate their extra electrons to the p-type atoms, which accept them. 
The atoms in the n-type material have extra electrons, while the atoms in the p-type material have electron holes - electrons missing from their outer rings.Īpplying electrical current to the diode pushes the atoms in both materials toward the junction area. 
To create an LED, the n-type material is negatively charged, while the p-type material is positively charged. Almost any two conductive materials will form a diode when placed in contact with each other - with a single p-n semiconductor junction between them. A diode is a device that allows current to flow in only one direction. As indicated by its full name - Light Emitting Diode - an LED is a diode that emits light. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
